Mark Lubberink, PhD, Scientific Advisor to MedTrace & Hendrik “Hans” Harms, PhD, Senior Scientist at MedTrace, are contributing to the November issue of Society of Nuclear Medicine and Molecular Imaging (SNMMI)’s Value Initiative Newsletter.
Lubberink & Harms elaborate on the challenges of using 15O-water positron emission tomography (PET) in clinical practice and how MedTrace aims to make 15O-water practically available. The article also expands on the analytical software aQuant that is under development.
Last March, the FDA approved MedTrace’s IND application and Phase 3 clinical trial RAPID-WATER-FLOW with the first patient enrolled on May 11, 2022.
Continue to read the full article below or find the November Newsletter from SNMMI here.

Author: Mark Lubberink, PhD, Scientific Advisor to MedTrace & Hendrik Harms, PhD, Senior Scientist at MedTrace.
15O-water positron emission tomography (PET) is widely considered the scientific gold standard for quantification of myocardial blood flow (MBF). It has been used for the technical validation of what is now the invasive gold standard, invasive pressure measurements (De Bruyne et al Circulation 1994), as well as to validate MBF quantification with SPECT (Ito et al EJNMMI 2003) or cardiac MRI (Tomiyama et al JMRI 2015). It has shown excellent diagnostic accuracy when compared to the invasive gold standard, outperforming both SPECT and CT angiography in the PACIFIC trial (Danad et al JAMA Cardiol. 2017).
Despite this, 15O-water PET has seen mainly research use and is not FDA approved for clinical use. One reason for this is that 15O-water has historically been challenging to use. It requires an on-site cyclotron to produce 15O and a radiochemistry facility and chemist to produce 15O-water. As 15O-water behaves exactly the same as regular water and is not retained in the myocardial wall, late uptake images do not show any contrast between the heart muscle and the blood pool, and dedicated software is required to visualize MBF. Any industry partners interested in bringing 15O-water to market had to provide both an improved and automated synthesis and analysis approach, which makes 15O-water commercially challenging.
MedTrace Pharma A/S is a Danish company that aims to make 15O-water practically available. It currently has an automated manufacturing device under Phase 3 investigation and is developing analytical software, aQuant. Its automated manufacturing device is in use clinically at Aarhus University Hospital in Denmark under regulatory exemption, where it has allowed a high patient throughput similar to that achievable with 82Rb (Hansen et al J Nucl Med 2021). As used under the regulatory exemption in Denmark, a rest and stress scan can be completed within 25 minutes, potentially allowing for more than 16 patients to be scanned during an 8-hour day.
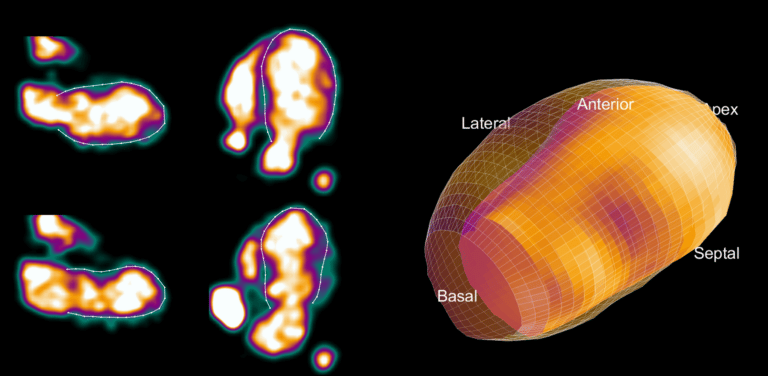
The key to working with 15O-water is to convert the images of 15O-water radioactivity concentration into contrast-rich images of quantitative MBF. The aQuant software under development is unique in that it has the ability to segment the arterial and venous blood pools first, and the segmentation of the left ventricular wall is done later. The blood pool segmentation is fully automated and identifies all the cavities of the heart and the large vessels with full reproducibility. A three-dimensional display of these regions, presented to the user for confirmation, shows a region including the venous side of the heart and a region including the arterial side of the heart.
After confirmation, the segmented blood pool is used to generate quantitative images showing MBF instead of radioactivity concentrations, and the original 15O-water images are discarded. The MBF images are then automatically rotated to the standard cardiac view and segmented as is standard for other nuclear MPI techniques. The clinician is finally presented with the rotated images to allow for visual inspection of the MBF distribution. In addition, quantitative polar maps and the MBF values of the standard 17 segments of the heart are displayed, and regions with abnormal MBF values are automatically highlighted. As currently being developed, it is expected that the entire analysis will be fully automated and only quality control will be required by a user.
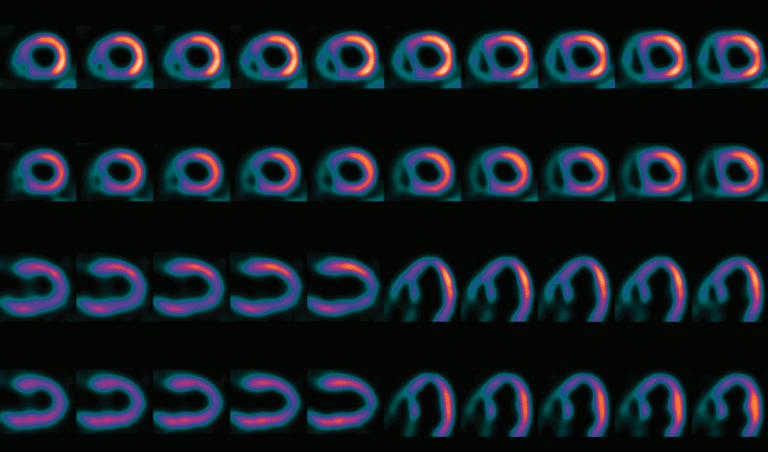
Finally, the lack of unprocessed uptake images requires a unique approach for the functional imaging, as there are no images of the heart muscle that can be used for calculation of volumes and ejection fractions. As being developed, aQuant makes use of a ‘negative’ image: shortly after injection, all 15O-water is present in the cavities in the heart, but not (yet) in the heart muscle. Creating an image of the beating heart during this timeframe shows the chambers of the heart contracting and relaxing, and this is the information needed to measure cardiac function.
In all, aQuant software has the potential to provide a fully automated analysis of quantitative MBF and cardiac function using 15O-water, with user input only required for confirmation and quality control. It has the ability to be fully integrated with a hospital’s file management system and internal network may be accessed through a web browser anywhere on a hospital’s internal network. If cleared by FDA, aQuant and the automated 15O-water manufacturing device may finally make the scientific gold standard 15O-water PET feasible for routine clinical use in diagnosing cardiovascular disease.